Authors:
- Mr. Avnish Kumar (AGM- Engg. & QA)- 12 years of industrial Experience {Quality of Ingot, Billets, Hot Rolling -Bars, Wires, Forged rounds}
- Mr. Sunil Sharma (GM- Engg. & Works)- 40 years of industrial Experience- Specially in melting {Melting: Arc Furnace, Induction furnace, AOD, LRF, VD, ESR,} & {Casting: Billet casting, Ingot casting & foundry Casting}
- Mr. Sourabh Gupta (Manager – Engg & QA)- 8 years of industrial Experience {Quality of Ingot, Billets, Hot Rolling -Bars, Wires, Forged rounds}
- Organization: Ambica Steels Limited; 51/2, Sahibabad industrial Area, Sahibabad Ghaziabad.
Corresponding Author Details:
Name: Avnish Kumar; Email Address: quality@ambicasteels.com,
Abstract
The Argon Oxygen Decarburization (AOD) process is a critical secondary steelmaking method predominantly used in the production of stainless steels and other high-alloy steels. This paper discusses the principles, operational steps, and chemical reactions involved in the AOD process. Furthermore, it outlines the advantages of AOD in producing high-quality stainless steels by controlling carbon content and minimizing oxidation losses of expensive alloying elements. Critical parameters for the economic control of AOD operation are also focused in this paper for attaining cost efficiency along with good quality of steel.

1. Introduction
Stainless steels are characterized by their corrosion resistance, achieved primarily through the addition of chromium and other alloying elements. Producing stainless steel requires precise control over carbon, sulphur, and nitrogen levels without excessive oxidation of valuable elements like chromium and nickel.
The AOD process, introduced in the 1950s, has become the standard method for refining stainless steels due to its ability to decarburize molten steel effectively while preserving these alloying elements. The process is typically performed after initial melting in an Electric Arc Furnace (EAF) or an Induction Furnace.
1.1 Advantages of the AOD Process
- High Chromium Recovery: Argon dilution reduces chromium
- Efficient Decarburization: Allows reduction of carbon to ultra-low levels (<0.03%) without significant loss of alloying elements.
- Flexibility: Capable of processing a wide range of stainless and specialty
- Lower Production Costs: High yield of expensive elements reduces overall production costs.
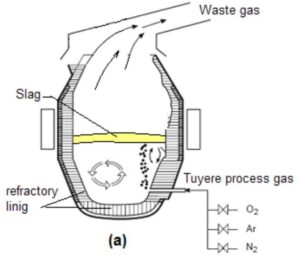
Fig 1. Image of typical AOD vessel
2. Overview of the AOD Process
The AOD process takes place in a specially designed converter known as an AOD vessel, which is a refractory-lined vessel capable of handling high-temperature molten metal. The process involves three major stages:
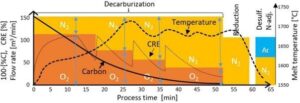
Fig 2. AOD process in one shot with process parameters
2.1 Carbon Boiling/ Oxidation/ Decarburization
- In this phase, a mixture of oxygen and inert gases (argon and/or nitrogen) is blown into the molten bath through submerged tuyeres or top lances.
- The purpose is to oxidize carbon selectively while minimizing chromium oxidation. For the same there is a parameter defined i.e. called CRE
- CRE: carbon removal efficiency is parameter which controls the effective carbon removal with lowering the oxidation of precious elements like Cr and other elements Mn & iron, Temperature & Blows ratio CRE generally varies from the 75% to 10 % in the whole process of decarburization.
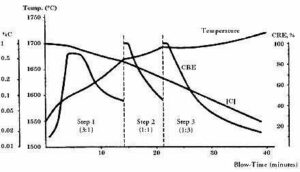
Fig 3. CRE & controlling parameters (temperature & blows)
- The basicity of slag is maintained high using lime (CaO) and dolomite (CaMg(CO₃) ₂) to facilitate the removal of sulphur in later stages.
2.2 Reduction
- After decarburization, valuable alloying elements like chromium and Mn, Iron may oxidize to some extent.
- A reducing agent such as ferrosilicon or aluminium is added to recover these elements from the slag.
2.3 Desulfurization
- Sulphur is removed by adjusting the slag composition and
- A reducing environment (low oxygen potential) is maintained, and lime is added to bind with sulphur to form CaS.
3. Chemical Reactions in the AOD Process & typical Slag making
3.1 Decarburization Reactions:
The main carbon removal reaction:
2C+ O2→ 2CO(g)
Chromium, Manganese & Iron also be oxidized:
4Cr+3O2→2Cr2O3(slag), 2Mn+O2→2MnO(slag),
2Fe+O2→2FeO (slag),
To minimize Cr, mn & iron loss, argon or nitrogen is mixed with oxygen to reduce partial pressure of CO and shift equilibrium.
3.2 Reduction Reactions:
Ferro Silicon is used as the reducing agent for the reducing the elements like Cr, Mn & Fe.
Recovery of chromium, Manganese, Iron from slag: 2Cr2O3+3 Si→4 [Cr]+ 3 SiO2,
2MnO+ Si→ SiO2+ 2Mn,
2FeO+ Si→ SiO2+ 2Fe,
3.3 Desulfurization Reaction:
For the better Sulfur reduction reaction, CaO/ Lime is required or high basicity reducing slag must be there with FeO less than 1%.
Sulphur removal:
[S]+ CaO→ CaS (slag)+[O]
3.4 Typical Slag composition:
Approximated typical slag composition majorly to be maintained in AOD is as below for the less lining erosion & better desulphurization:
Table 1: Typical composition of the slag
|
%CaO |
%MgO |
%SiO2 |
%FeO |
%MnO |
%Cr2O3 |
%R2O3 |
| 46% | 12% | 30% | Below 1% | 2% | 2% |
7% |
4. Equipment and Operational Considerations
- Converter Design: Typically, cylindrical with a conical bottom containing tuyeres for gas injection.
- Gas Control System: Precise flow rate control for oxygen, nitrogen and inert gases is crucial for process stability.
- Refractories: Basic refractories (MgO-CaO based) to withstand slag chemistry and high temperatures. Cost of the refractory can be managed by increasing life.
5. AOD Refractory Life Controlling Parameters
Refractory lining is basic in nature i.e. dolomite very expensive, and frequent replacement increases operating costs. Longer lining life reflects lower refractory cost per ton of steel. The following is the image of the respective lining zone wise & the controlling parameters for maintaining good lining life:
Refractory limning is optimized for the efficient utilization of refractory bricks, for the same reasons the vessel is divided in to the five nos of Zones: Bottom, metal hot zone-Tapping Side (less turbulence & erosion), metal hot zone-Tuyere Side (High turbulence & high erosion), Slag Zone (Chemical erosion high). Most erosive zones have high quality longer length refractory bricks & normal quality bricks with lesser lengths can be used in less -erosive zones.
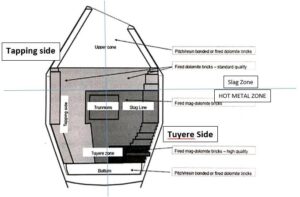
Fig 4. Zones in the AOD & lining quality as per Zone
Majority of factor affecting the lining life of refractory are summarised in below table with quantification.
Table2. total nos. of bricks its length wise used in 30 MT AOD
| SN | Size-length | lining | cone | bottom |
| 1 | 55 | 150 | ||
| 2 | 50 | 110 | ||
| 3 | 45 | 70 | ||
| 4 | 40 | 60 | ||
| 5 | 35 | 85 | ||
| 6 | 30 | 110 |
| 7 | 25 | 665 | |||
| 8 | 20 | 150 | |||
| 9 | 15 | 240 | |||
| 10 | Tuyer-65 | 2 | |||
| 11 | 30 | 230 | |||
| Total | 1250 | 390 | 230 | 1870 |

Fig5. Vertically cut – Elevation View of AOD -30 MT capacity, Showing Economic
arrangement of different length & quality bricks as per its erosion pattern wise.
Table3. Factors affecting the AOD refractories work lining life
| SN | FACTOR | PARAMETERS | EFFECT OF FACTORS |
| QUALITY OF MATERIAL IN AOD: | |||
| 1. | Quality of bricks | a) %CO2 < 1
b) % MgO and shape >65 /spherical c) % FeO < 1 d) %ZrO2 up to 1 |
If %MgO will be less in quantity and not been used proper shaped it will not be able to make MgO-FeO compound layer over the surface of lining. |
| 2. | Quality of Lime & dolomite flux | a) CO2 – 1% to 1.8%
b) LOI- < 3% c) Temp pick up in 2 min – 20 to 60o |
High CO2 and LOI reduces the CaO content, temperature pick up tendency along with poor CRE result. |
| CHEMICAL REACTION BASED EROSION: | |||
| 3. | Initial Slag Practice | Basicity > 1.20 | For nullify the effect of Initial Si opening, the basicity must be maintaining just above the critical value of given. |
| 4. | Decarburising Slag practice | Basicity > 1.00 | As we know the decarburising Slag contains all amphotaric oxides like Cr2O3, MnO, FeO so the nullify these oxides the Flux must be added by |
| keeping Basicity 1.2 and addition Must be completed in First blow as possible. | |||
| 5. | Reducing Slag Practice | Over all slag basicity ≥ 1.50 | If Overall Slag basicity is lesser than the Optimum value, then erosion is faster. |
| THERMAL SHOCK PARAMETERS: | |||
| 6. | Preheating of Lining | 24 hrs cycle must be followed
1st soaking at 650- 700 2nd soaking at 1100 must |
For 1.reducing chilling; 2.maintain permanent linear change; 3.improving hot compressive strength of bricks;
4.reduces spalling also. |
| 7. | Working Temperature | Temp < 1700o C | High temperature leads to shrinkage of hot face of bricks thus pockets create between bricks. |
| 8. | Heat Process Time | Reduced as much as possible | Reduce heat process time, the work lining life will automatically increase in nos. of heat. |
| 9. | Holding Time of Vessel | Must be avoided | High holding time generate thermal stresses just because of thermal shocks which results Spalling defect in bricks. |
| 10. | Knurdle Formation | Improper cooling / High flow of Argon and high temperature at tip of tuyere | Knurdle is deposited iron oxide and other oxides over the surface of the tuyere pipe. Most of the time it creates the turbulence in the nearby area of tuyere and creates cobble stoning in bricks. |
| OPERATIONAL- GEOMETRY PARAMETERS: | |||
| 11. | Hot face
distances between tuyere bricks |
Hot face distance > 16” | Less distance between the hot faces of tuyere leads towards high erosion. Keep in mind during the three tuyere operations. |
| 12. | Tuyere Angle | Between 57º to 67º, Set nearby centre of vessel can be adjustable 1” to 3” by seeing erosion | If tuyere angle is not set proper then the Erosion will be higher in tuyere pad as well as in those areas, where it should not be. |
| 13. | Tuyere centre | 6-8” shifted towards tuyere side | It will not hit Tapping side where lining thickness is less as per economical design |
| 14. | Back tilting of AOD | 5º to 8º angle from perpendicular | In Side- tuyere AOD the blown gas creates big elliptical current and small elliptical current. For lowering the velocity of small elliptical current which is nearer to tuyere, back tilt of AOD must be done. |
| 15. | AOD converter d/h ratio | D / H > 1.8 | If not maintained, then the area per turbulence will increase and erosion will be higher side. |
7. Discussion
Improved CRE (Carbon Removal Efficiency) will reduce the oxidation of elements such as chromium (Cr), manganese (Mn), and iron (Fe) & with reduced gases like Oxygen & Argon consumption. As a result, ferro silicon consumption will decrease, along with the requirement for fluxes like lime and dolomite. Better slag practices, with optimized and lower flux usage, will also help extend refractory lining life and reduce associated costs. This efficient approach enables the production of cleaner steel with minimal metallurgical waste (slag), making the process more economical and sustainable.
Intelligent Refining Systems utilize advanced control and monitoring technologies to enhance CRE, reduce oxidation losses, and improve overall metallurgical efficiency.
Case Study: In general, CRE of process appears 25-35%, it can be increased up to 50% & there is always scope of improvement of CRE. If 10% CRE is improved in 25 MT AOD, which consumes approx. 1000Kg FeSi, 1000NM3 Oxygen, 2000KG Lime 1200 KG Dolomite. Then the cost saving in consumable will be = 2rs/Kg minimum on a small production of 5000MT Stainless Steel per month production, 1Cr Rs./month can be saved.
Refractory lining life is another most important parameter which increases the operational cost of the manufacturing because dolomite refractory bricks cost is huge. By using the proper quality of bricks, fluxes & good operation practices life of refractory can be increased thus cost of the refractory can be minimized.
Case Study: By maintain good practices of melting as per the table 2, 12lac rs./month can be saved on 5000 MT/month capacity plant.
8. Conclusion
The AOD process revolutionized the production of stainless steels by allowing precise control over carbon content and alloying elements. AOD process can be more cost-effective by improving two major operational parameters – CRE & Refractory Lining Life.
References
- Turkdogan, T. “Fundamentals of Steelmaking.” The Institute of Materials, 1996.
- Fruehan, J. “The Making, Shaping and Treating of Steel: Steelmaking and Refining Volume.” AISE Steel Foundation, 1998.
- Peters, M. C., and Timpe, G. L. “Argon-Oxygen Decarburization of Stainless Steel.” Journal of Metals, 1968.
